Medical devices and implants face a unique set of challenges inherent to these types of delivery systems that must be addressed to ensure optimal performance. The complete system is affected by environmental conditions (moisture, temperature changes, VOCs, etc.) that can result in degradation of the drug substance, reduced potency, and ultimately deliver sub-therapeutic levels to patients. Aptar’s material science-based offerings create customized, highly-engineered solutions to overcome these challenges and ensure we all keep our promises to patients around the world.
Implants
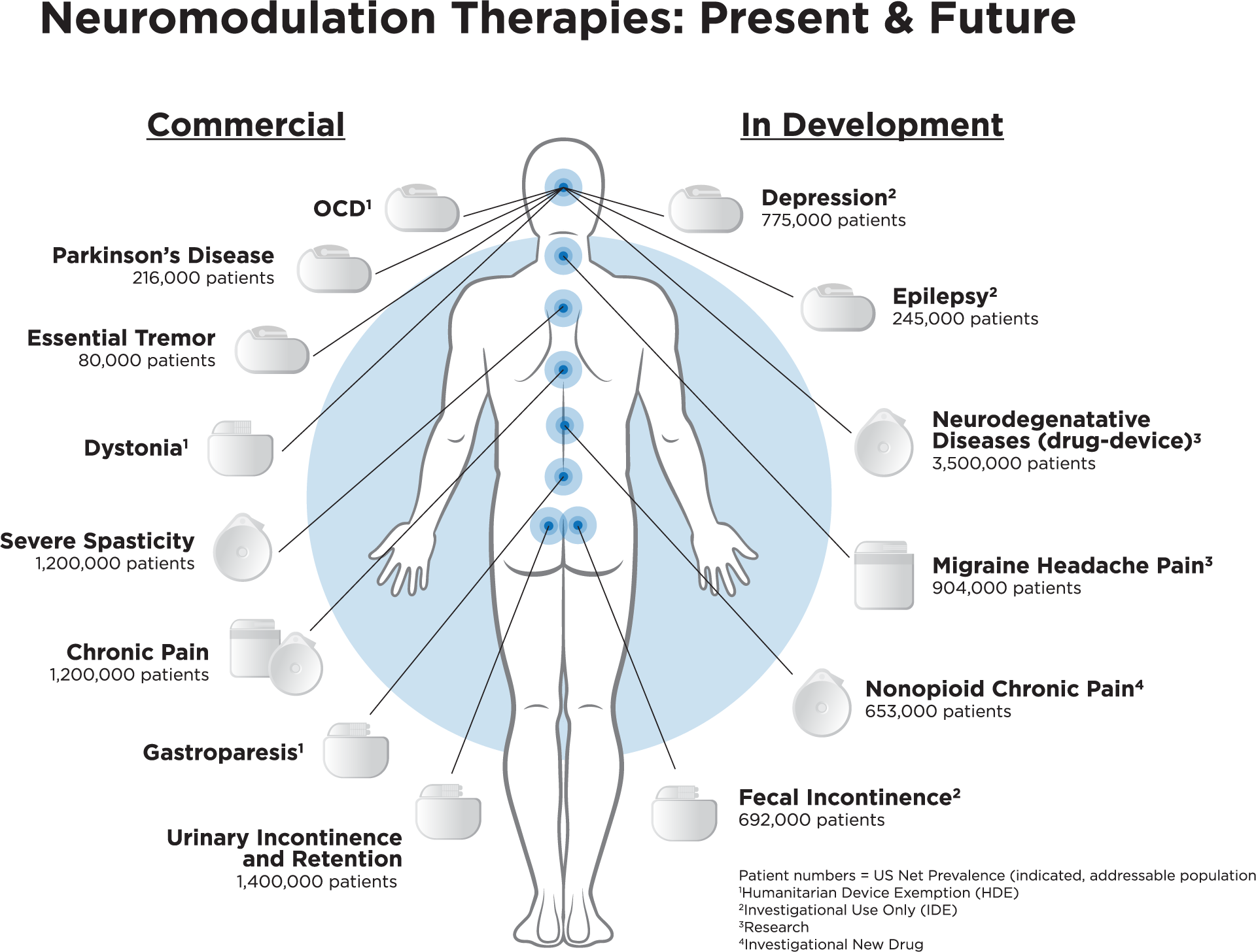
Implantable devices have to perform as intended 100% of the time. Pacemakers, Implantable Cardioverter Defibrillators (ICDs), Cardio Resynchronization Therapy devices, and neuromodulators, are lifesaving devices that require unique protection from environmental exposure that can affect the functionality and lifespan of the device. While plastics in these devices act as insulators, they can also emit moisture, as do the printed circuit boards (PCBs) inside them. These devices are often purged with inert gas/hermetically sealed in an effort to protect the electronic components from moisture-induced corrosion that can lead to decreased battery life.
Aptar Active Material Science offers a fit for purpose solution to deliver the precise microclimate needed to protect implantable devices and drug delivery systems. The technology controls the internal atmosphere surrounding the electronic components, delivering optimal moisture protection and minimizing circuitry corrosion to extend battery life.
Activ-Film™ Material
Activ-Film™ material offers a custom-engineered, fully-integrated solution that adsorbs moisture over the life of the device and sterilizes medical devices. The film can be heat staked into the medical device itself or applied to outer packaging to ensure optimal device performance. Using innovative active polymer technology, the film can adsorb tailored amounts of moisture, scavenge oxygen or VOCs, or provide a combination of active protection. Activ-Film™ material can be precision die cut (tolerance +/- 0.01 mm) to accommodate any device design.
Active Molded Component
Combining our formulation and molding expertise, Aptar can develop a customized active component to create an ideal microclimate inside the medical device, helping to ensure optimal performance. Active molded components can be seamlessly integrated to deliver extended use life by assuring protection from environmental factors that can degrade device efficacy. These components often replace an existing plastic part in the medical device with an active component that controls the head space environment. Insertion of the part can be automated without breakage. The result is maintained physical function while incorporating the performance of the 3-Phase Activ-Polymer™ technology.
Why Aptar Active Material Science?
Expand market access to ICH Zones 3 and 4
- Enhanced stability and efficacy due to moisture adsorption and oxygen scavenging
- Extends shelf life over traditional options
- One solution to fulfill global markets
Minimize failure risk and maintenance of Human Implantable Stimulators and e-MD
- Extends battery life by minimizing circuitry corrosion
- Electrical insulation – low dielectric constant
Versatile solution
- Customized formulations to meet device-specific needs
- Customized adsorbing and releasing kinetics and capacities
- Specific VOCs control
Expedite time to market
- Avoid redesign costs by utilizing empty headspace of device
- Solutions ideal for reduced headspace
- Highest adsorbing capacity (up to 70% of the volume/mass)
- ISO 8 parts production and sterilization for aseptic packaging
- Low particulates with COC based formulation
- ISO 13485 / ISO 9001 / 21CFR820.30